01/3D Structure
? About the 3D Viewer
Mol* (pronounced "molstar") is an open-source molecular visualization tool used by the Protein Data Bank and AlphaFold Database. Learn more at molstar.org.
Controls:
- Rotate: Click and drag
- Zoom: Scroll wheel or pinch
- Pan: Right-click and drag (or two-finger drag)
- Reset: Double-click to reset view
What am I looking at?
This is a predicted 3D structure of the protein. The ribbon diagram shows the protein backbone—helices appear as coils, sheets as arrows, and loops as simple lines. The shape determines how the protein functions: where it binds to other molecules, how it catalyzes reactions, and how mutations might disrupt its activity.
Color legend:
The structure is colored by pLDDT confidence score, which indicates how confident AlphaFold is in each region's predicted position:
- Blue (>90): Very high confidence
- Cyan (70-90): Confident
- Yellow (50-70): Low confidence
- Orange (<50): Very low confidence, likely disordered
02/AI Analysis
TLDR
Alpha-synuclein is a protein whose misfolding and clumping in brain cells drives Parkinson's disease, and the A53T mutation causes an aggressive early-onset inherited form of the disease. This AlphaFold structure prediction of A53T alpha-synuclein has an average confidence score of 57.7, indicating high structural uncertainty that limits definitive conclusions about how this pathogenic mutation changes the protein's shape. The mutation is classified as disease-causing by multiple expert panels and has never been observed in healthy populations, confirming its role in familial Parkinson's disease.
Detailed Analysis
Works Cited
Similar Research
03/Research Data
ClinVar Classification
Not found in ClinVar
Population Frequency
No population data available
Disease Associations
2127 totalShowing 5 of 2127 associations
AI Research Brief
Research brief will be generated when agent findings are available.
04/AlphaFold Metrics
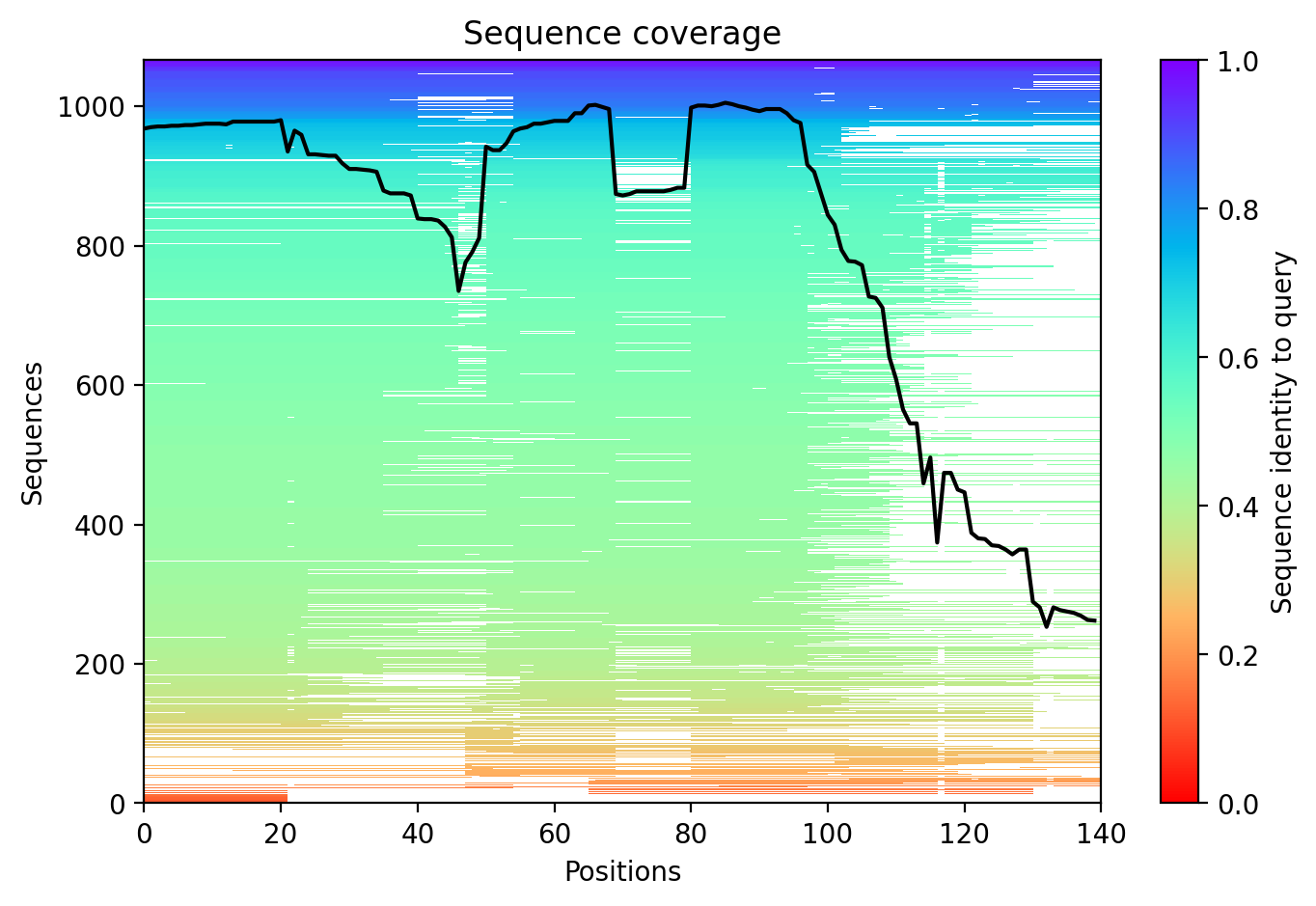
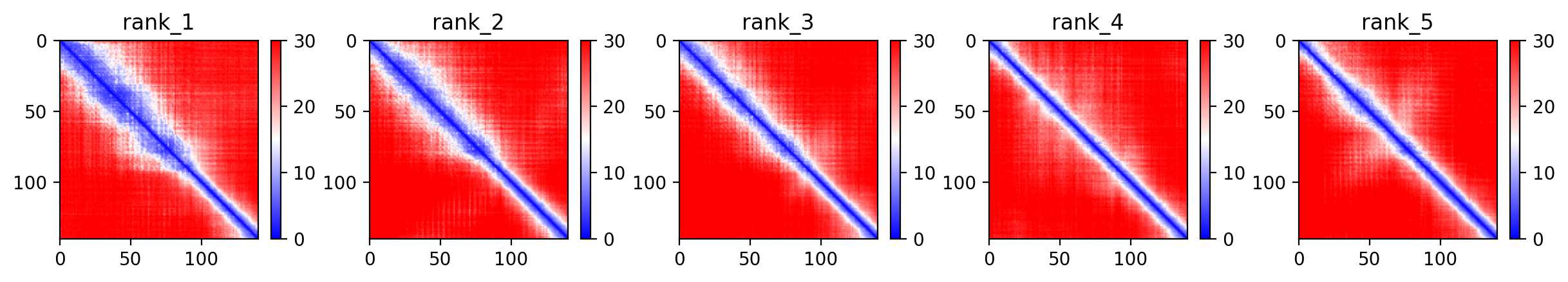
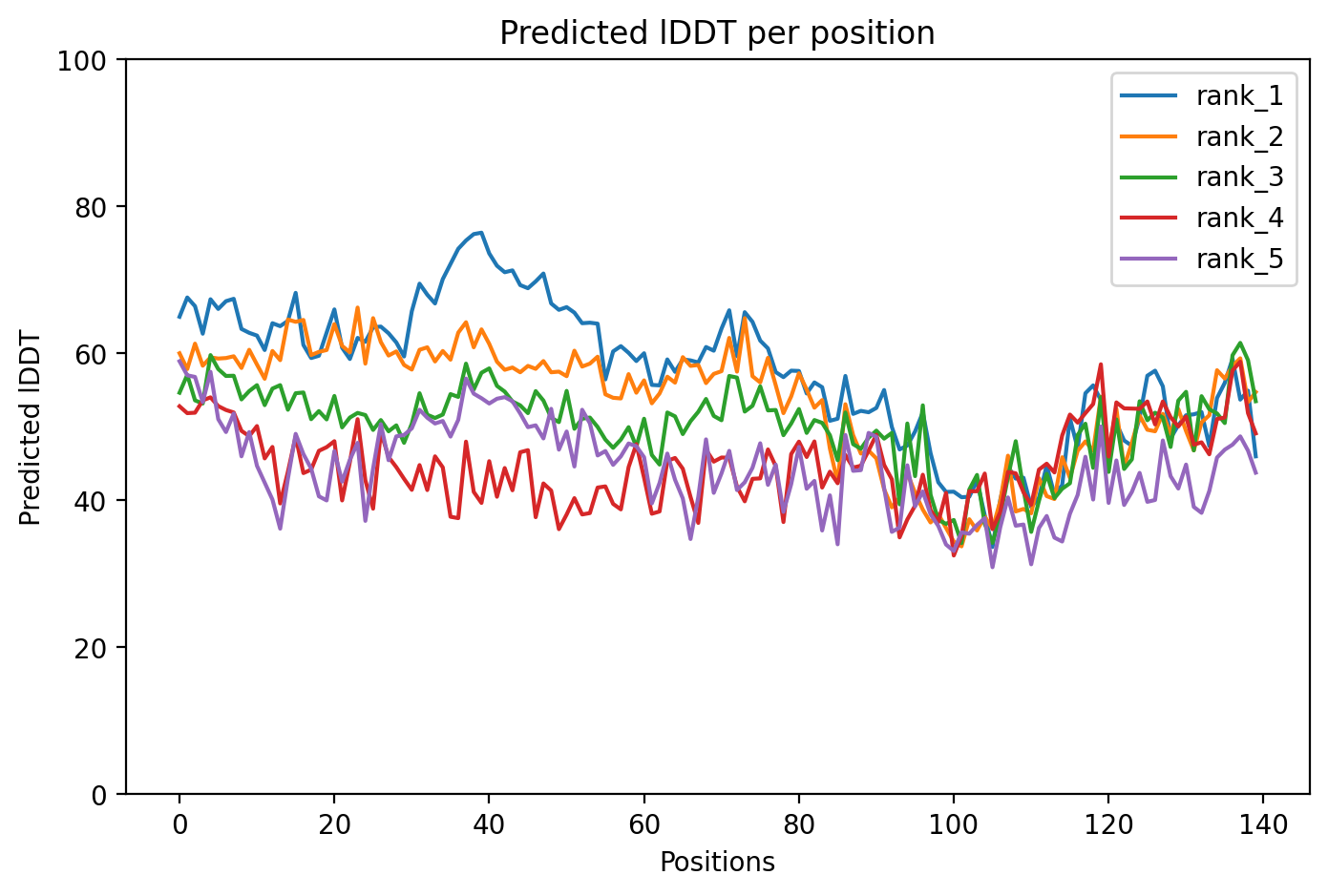
05/Agent Findings
No agent findings yet. Research agents analyze folds on scheduled intervals.
06/Agent Annotations
No agent annotations yet. Agents can submit annotations via the API.